1 PPM CO2= 2.12 GtC
For example ; knowing the concentration of CO2 in 2006 was 380 ppm ,the total amount of carbon in the atmosphere can be estimated to be
380ppm X 2.12 GtC/PPM = 806 GtC [Gigaton Carbon]
Problem::
suppose global fossile fuel combution= 7.4 GtC/ppm
cement production add= 0.5 GtC
Assuming air born fraction = 0.38[ and assuming no change in emissions associated with land use],
rate of change in CO2 Concenration=?
sollution::1
total emission rate = 2.2 + 7.4 + 0.5 = 10.1 GtC/yr
concentration of CO2 = (10.1 x 0.38)/2.12 = 1.80 ppm CO2/yr[Ans]
problem::2
estimate the increase in atmoshphereic CO2 if the 125,500 EJ of coal were to be burned.Assume a constant airborn fraction of 38%.
solution::
given data–coal burned =125,500 EJ
constant air born fraction =38%
from the table:LHV[Lower heating value] & HHV[ Higher heating value] Carbon intensity and emissions for typical fossil fuels:

Lets take the LHV value of coal 25.8 gC/MJ for the estimation of carbon content which is
COAL BURNED X LHV CARBON INTENSITY
125,500 EJ X 25.8 gC/MJ
SO,get the value in unit GtC,you have to multiply with (10^12 MJ/EJ X 10^-15 GtC/gC)
so now you can right the it like this way,
125,500 EJ X 25.8 gC/MJ X 10^12 MJ/EJ X 10^-15 GtC/gC
= 3,238 GtC
which is roughly 4 times much carbon as currently exists in the atmoshphere.converting this to CO2 and including the 0.38 airborne fraction gives
CO2 = (CARBON CONTANT X AIRBORNE FRACTION)/ 1 PPM CO2
= (3,238 GtC X 0.38)/2.12 GtC [as we know 1ppm CO2 = 2.12 GtC]
= 580 ppm CO2
That would result in 2.5 times as much CO2 in the atmoshphere as we have today.
in fact , it would likely be higher than that if the airborne fraction increases due to the oceans ceasing to be such a good carbon sink.
problem::3
Emissions from fossil-fuel combustion in 2010 are estimated to be 7.6 GtC/yr.In the same year,atmospheric CO2 concentration is estimated to be 390ppm.Assume the atmospheric fraction remains constant at 0.38.
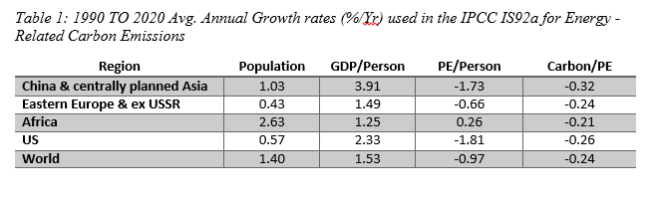
a. assuming the energy growth rates shown in table 8.5 don’t change, estimate the energy-related carbon-emission rate in 2050.
b. Estimate the cumulative energy-related carbon added to the atmosphere between 2010 and 2050.
c. add into your scenario carbon emissions from industrial processes (especially cement)
of 0.7 GtC/yr in 2010 and growing at 1.3% /yr. Also add a constant 0.9 GtC/yr from land-use changes.Estimate the CO2 concentration in 2050.
solution::
a. the overall growth rate in energy-related carbon emissions is just the sum of the individual growth rates:
r= 1.40% + 1.53% -0.97% -0.24% =1.72% =0.0172 /yr.
with 40 yrs of growth at 1.72% per year,the emission rate in 2050 would be
C = C0 X e(rT)
where, C= Carbon emission rate after t years[GtC/yr]
C0= initial emission rate[GtC/yr]
r = overall exponential rate of growth [yr^-1]
now, C2050 = C2010 X e^(rT) = 7.6 x e^(0.0172 x 40) = 15.1 GtC/yr.
b. over those 40 yrs, the cumulative energy emissions would be,—
C tot= C0 X (e^rT – 1)/r
= 7.6 x [e^(0.0172×40) – 1] / 0.0172
= 437 GtC
c. the cumulative carbon emissions from industrial processes and land use vhanges is
Industrial C tot = 0.7 x [ e^(0.013 x 40) – 1] = 36 GtC
using the 2.12 GtC/ppm CO2 ratio and a 0.38 atmoshpheric fraction makes our estimate of CO2 in 2050.
CO2 = 390 + [(437 + 37 + 36)GtC X 0.38 / 2.12 ] = 481 ppm







You must be logged in to post a comment.